 In this test, first, we take the food sample. We can differentiate between reducing and non-reducing sugars by the following test: Benedict’s Test 
Test to distinguish reducing vs non-reducing sugars Note that, polysaccharides such as starch are non-reducing sugars. So, sucrose does not contain a free carbonyl group. Hence, the carbonyl groups of both monosaccharides participate in the glycosidic bond. This is because the combination of glucose and fructose forms it. 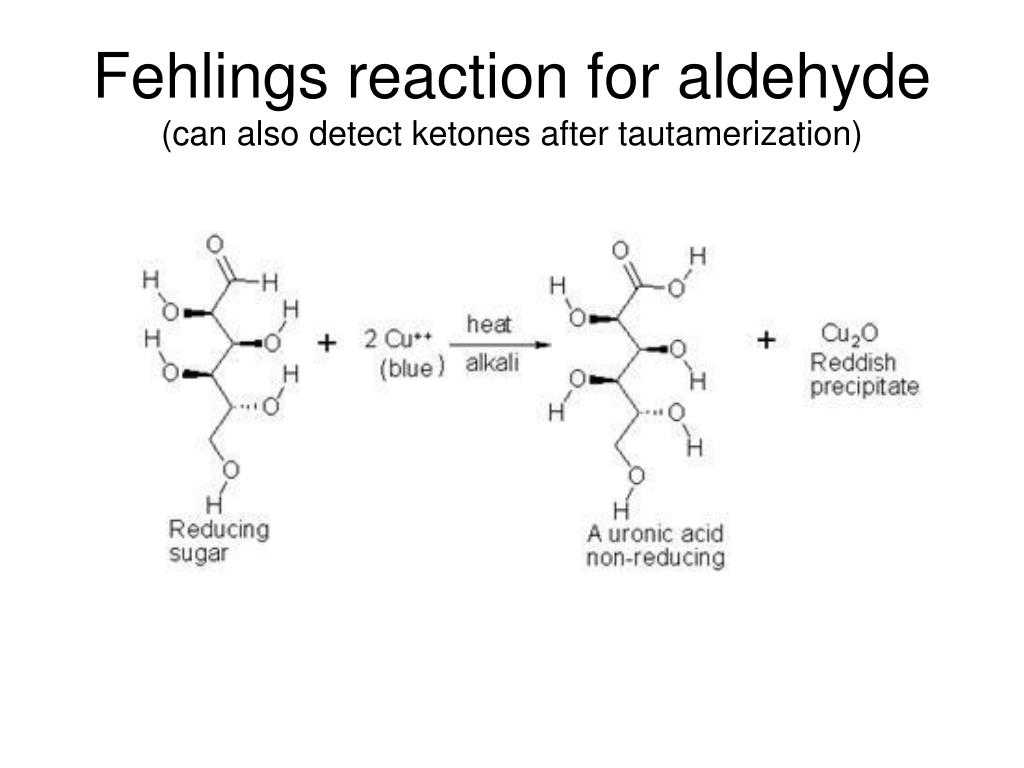
They do not contain anomeric carbon attached to the hydroxyl (-OH) group.įor instance, sucrose is a disaccharide but it is a non-reducing sugar. Non-reducing sugarsĬarbohydrate which does not contains free aldehyde and ketone group is known as non-reducing sugars. Some disaccharides are also reducing sugars such as lactose. The anomeric carbon is that which is derived from the carbonyl group and contains two oxygen substituents.Īll monosaccharides are reducing sugars. However, reducing sugars contain free anomeric carbon. 
They can donate electrons to other compounds and cause the reduction of other compounds. Reducing sugars contain free aldehyde and ketone groups. Reducing sugars Non-reducing sugars Sugars that have a free aldehyde or ketone group and can reduce other compounds Sugars that do not have a free aldehyde or ketone group and cannot reduce other compounds Common Examples: glucose, fructose, maltose Common Examples: sucrose, lactose, cellulose Positive result with Tollen's reagent (forms a silver mirror) Negative result with Tollen's reagent (no reaction) Color changes are observed in Benedict test no color change in Benedict test They can be used as precursors in millard reaction Not used as precursors What are reducing and non-reducing sugars? Reducing sugars 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
